 This means that they do not have any sharp melting point and are non-crystalline. But both are polar.Īmorphous solids are amorphous. This means that they have no definite edges. Amorphous solids are softer and more pliable than crystalline ones. The former has a sharp melting point and is brittle. The difference between crystalline and amorphous is mainly based on the structure. Amorphous solids are often more dense than crystalline ones. An amorphous solid has no definite or characteristic heat of fusion. It is characterized by a sharp melting point and a definite melting temperature. They are anisotropic.Ī crystalline solid is rigid and has a regular geometry. Unlike crystalline solids, amorphous solids do not show long-range order. Amorphous materials do not have a defined shape. They are homogeneous, asymmetrical, and have no sharp edges. They do not have definite melting points. They have no definite defining temperature.Īmorphous solids are characterized by irregular breakage. The amorphous solids have a wide range of melting and have no characteristic shape. Amorphous materials have no sharp melting point and lack definite structure. The difference between crystalline and amorphous materials is primarily determined by the degree of ordering. This means they can be easily broken and are often not re-usable for industrial processes. Amorphous materials do not have definite melting points. Crystals have definite melting points and their constituents are arranged in an orderly fashion. The difference between crystalline and amorphous material is based on their melting points and their cleavages. The distinction between amorphous and crystalline solids is very useful when describing the properties of different materials. Because of this, they are impossible to identify by their structure as a crystalline one. They are identical in all their properties along all their axes. In addition, they do not have any characteristic geometry. Amorphous solids lack distinct edges and are therefore non-crystalline.Īmorphous solids lack a long-range arrangement of atoms. Despite their name, amorphous materials do display an orderly arrangement of atoms that may extend up to a few Angstrom units. 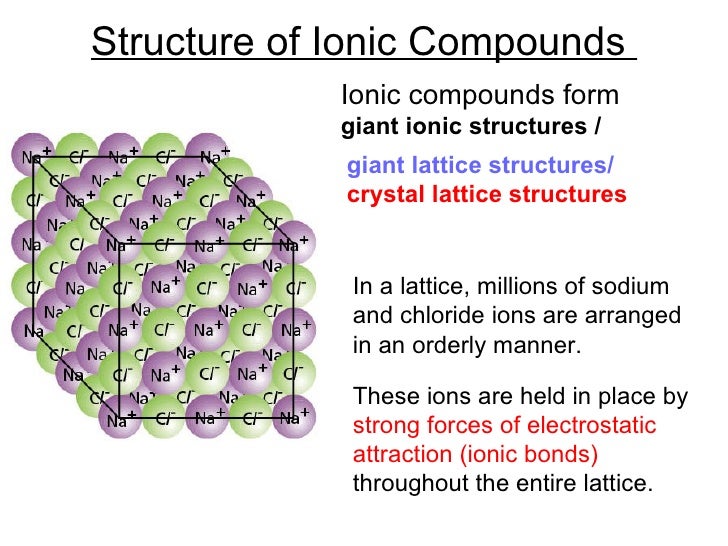
They also do not have a fixed melting point. This makes amorphous compounds highly rigid and have irregular surfaces.Īmorphous solids are non-crystalline substances that do not possess a characteristic geometrical arrangement. In contrast, an amorphous compound has a short range of ordered molecules and an irregular arrangement of its atoms. A crystalline solid has a long range of ordered molecules and a sharp melting point. The fundamental difference between crystalline and amorphous compounds is the arrangement of their constituent atoms. 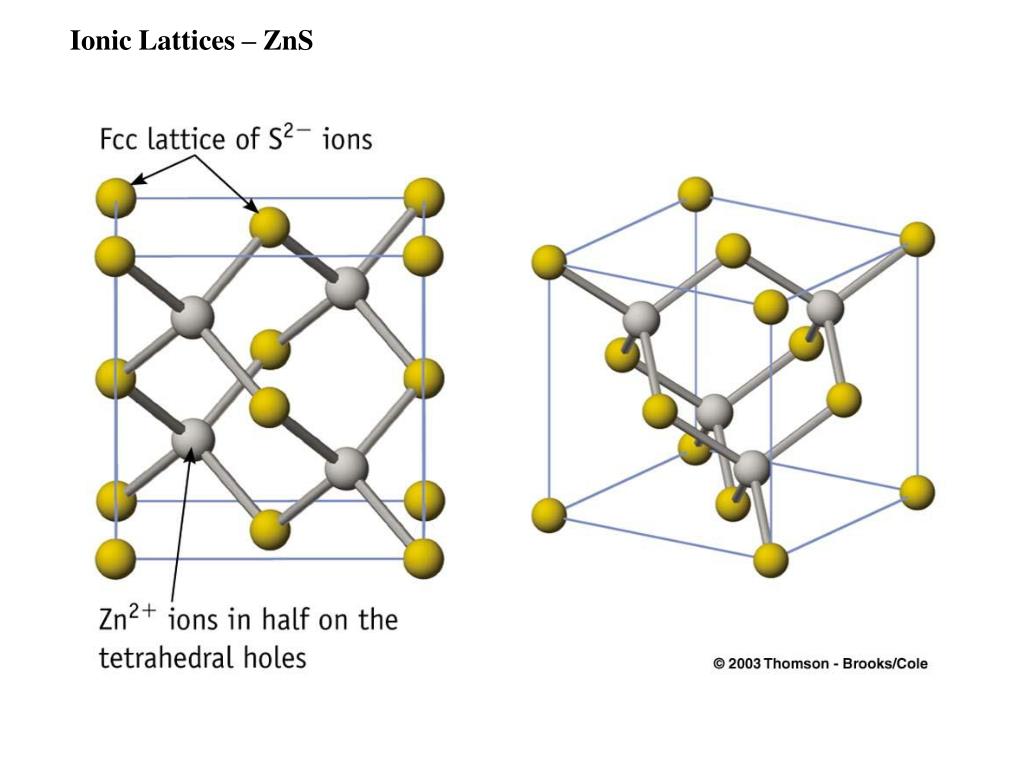
Send Determining The Difference Between Crystalline & Amorphous Solids Get Your Quote FAST and Starte Researching Today! Your Name: the angle between edge b & c is ( ?), and a & c is B and that of between a & b is v. the a,b & c may not be mutually perpendicular A unit cell has three edges a,b & c and three angles ( ?),B & V between the respective edges This gives them a very high hardness but also gives them a low toughness as they can be easily fractured along certain crystal planes.2.) The following characteristics define a unit cell. Unlike ionic and metallic bonding, covalent bonding is directional and as a result, the atoms in many covalently bonded ceramics are arranged symmetrically to give a highly ordered structure. 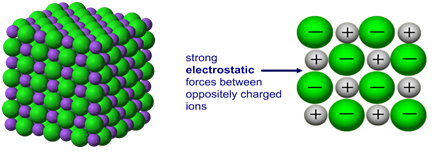
The high energy of covalent bonds makes these ceramics very stable with regard to chemical and thermal changes. The atoms in these ceramics are arranged so that each pair of nearest neighbour atoms forms a chemical bond by sharing a pair of electrons. Covalent bondingĬovalent bonding is found in many ceramic structures such as SiC, BN and diamond. The nature of the ceramic depends on the size of the ion charges and the size of the ions.įor example, the structure shown left is the rock salt structure, such as that for NaCl. The ions pack into a regular arrangement. Atoms have unlike electrical charges, making them ions, which create an electrostatic attraction between atoms. Ionic bonding is found in many ceramic structures such as NaCl, MgO and Al2O3. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
